CESMA - Design and Synthesis of Analgesic Molecules
Leader: Sylvie Ducki, prof.
Research themes
The principal objective of CESMA is to develop new molecules having analgesic activity in collaboration with the partners of Cluster of Excellence Analgesia Partnership (www.analgesiapartnership.com).In order to achieve our goals, we have identified two approaches:
- Rational approach: The therapeutic target is identified by the pharmacologists and validated by specific studies. CESMA will endeavour to design and synthesize organic molecules aimed at these targets. The objective will be to use these small organic molecules as tools in order to better understand the implications of the targeting and validate the proof of concept and will prove that the identified targets are exploitable for the development of analgesic drugs.
- Traditional approach: Many traditional plants are used in the treatment of pain. The efforts of research will involve the identification of such plants and their extraction in order to isolate the active ingredients. These molecules will be subjected to pharmacological studies to elucidate their modes of action, and could result in the emergence of new therapeutic targets.
People
- Robin AUGUSTE (PhD student)
- Mathilde BARD (Master)
- Guillaume CARDIN (PhD student)
- Corentin CHARBONNIER (Master)
- Marie CHASTANET (Master)
- Norberta DELPORTE (Master)
- Antoine DUMEIGE (Chef de projet Innopain)
- Laurie GONTHIER (Engineer)
- Vassili PRUDHOMME (PhD student)
- Marion ROUBY-LEROUX (Master)
- Ghislaine TSONA (PhD student)
- Rachida ZOUIBAA (Master)
Expertise
The research of CESMA is at the interface of chemistry / biology and scientific skills include organic synthesis and molecular modeling, drug design, fundamentals in neuroscience and strong interactions with bio-scientists.
Project TREK
Description of project
Potassium channels TREK-1 were identified as an important target in the perception of the pain. [Alloui 2006, Noel 2009] Indeed, they showed that the treatment of TREK-KO animals with analgesics of reference did not produce any analgesic effect whereas the activation of this channel led to an analgesic effect, thus validating the therapeutic target. A screening of molecules known as activators of TREK-1 was carried out by our team and allowed us to identify a lead molecule, able to activate TREK-1 channels (shown by electrophysiology) and having an analgesic effect in vivo (test of the acetic acid). Analogues of this lead were conceived then synthesized in order to identify the pharmacophore of the lead. These molecules were the subject of a pharmacological evaluation and a molecule emerged with a promising pharmacological activity.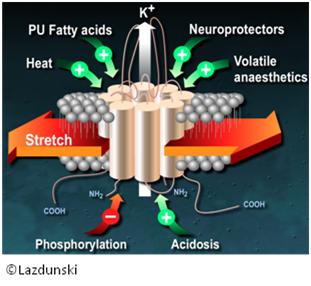
Publication
Ducki, S. ; Rodrigues, N. ; Bennis, K. ; Eschalier, A. « Composés anti-Douleur », FR1162564, French patent deposited on December 30, 2011.Grants
Bourse Innovation du Conseil Régional d’Auvergne / FEDER (2009-2011) Bourse Novartis (2011) Bourse MESR (2011-2014)
Academic and industrial collaborations
Dr Jérôme Busserolles et Pr Alain Eschalier - Equipe « Pharmacologie fondamentale et clinique de la Douleur » de l’Institut NeurolDol (UMR1107) à l’Université d’Auvergne, Clermont-Ferrand. Dr Florian LESAGE – Equipe « Physiologie moléculaire et physiopathologie des canaux ioniques » de l’Institut de Pharmacologie Moléculaire et Cellulaire (UMR7275) à Valbonne Sophia Antipolis. Dr François Caussade – ANS Biotech, Clermont-Ferrand.
Project PDZ
Description of project
Studies have shown that the interruption of the interaction between the PDZ protein PSD-95, and the serotonin receptor 5-HT2A by a peptide containing the 9 C-terminal residues of the receptor 5-HT2A (thus preventing the interaction between PSD-95 and 5-HT2A) is accompanied by an analgesic effect in the mice suffering from neuropathic pain [Pichon 2010]. Consequently, it becomes interesting to design and synthesize small inhibiting molecules of PDZ domains.
The problem lies in the selectivity of inhibition. Indeed more than 250 PDZ domains are encoded by the human genome [Ducki 2009]. We thus chose a general approach which aims: 1) to explore the similarities and differences between PDZ domains, 2) to design and synthesize small specific inhibiting molecules.
Our approach thus consists in evaluating bi-ligands interaction with PDZ domains, these bi-ligands constituted by a part interacting with the S0 site (Our choice was made on an indole described in the literature [Fujii 2003, Vogrig 2011] for its interaction with the S0 site) and the other part exploring the site S1 (linker Cn + acid amino). A first series of 16 molecules was prepared and evaluated (linker: C2, C4, C6 + amino acid: Ile, Lis, Trp, Tyr) [Boucherle 2011].
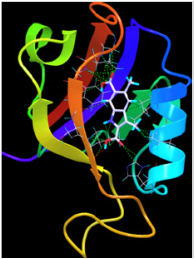
Publications
Boucherle B, Vogrig A, Deokar H, Bouzidi N, Ripoche I, Thomas I, Marin P, Ducki S. (2011) Synthesis and evaluation of bidentate ligands designed to interact with PDZ domains. Bioorg Med Chem 19:4346.Ducki S, Bennett E (2009) Protein-protein Interactions : Recent Progress in the Development of Selective PDZ Inhibitors. Current Chem Biol. 3:146.
Vogrig A, Boucherle B, Deokar H, Thomas I, Ripoche I, Lian LY, Ducki S. (2011) NMR evaluation of interactions between substituted-indole and PDZ1 domain of PSD-95. Bioorg Med Chem Lett 21:3349.
Grants
ANR-PCV « PDZ-CANPAIN » (2009-2012) Thématique Emergente du Conseil Régional d’Auvergne / FEDER (2009-2012) Postdoc PRES Clermont-Université (2010-2011)
Academic and industrial collaborations
Prof Lu-Yun Lian, Center for Structural Biology, University of Liverpool UK
Prof Christine Courteix, Equipe « Pharmacologie fondamentale et clinique de la Douleur » de l’Institut NeurolDol (UMR1107) à L’Université d’Auvergne, Clermont-Ferrand.
Prof Philippe Marin, Equipe « Sérotonine et Physiologie » de l’Institut Génomique Fonctionnelle (UMR5203), Montpellier.
Project CGRP
Description
CGRP is a neuropeptide, composed of 37 amino acids, which has several physiological functions and which is widely distributed in the peripheral central nervous system. Thus the neurons containing CGRP modulate the function of immunized, respiratory, endocriniens, gastro-intestinal, musculo-skeletal and cardiovascular systems. The presence of CGRP and its receptor in brain tissues is closely associated with the modulation of the transmission of pain.The objective is to design a new family of potential tricyclic antagonists of the CGRP receptor which plays an important role in the modulation of the inflammatory pain.
Grants
Bourse Novartis (2009) Postdoc PRES Clermont-Université (2010-2012)Academic and industrial collaborations
Prof Alain Eschalier, Equipe « Pharmacologie fondamentale et clinique de la Douleur » de l’Institut NeurolDol (UMR1107) à L’Université d’Auvergne, Clermont-Ferrand.Project Guiera
Description
This project aims at studying the chemical composition and the pharmacological activity of a plant used in the Western African traditional pharmacopeia, and more particularly prescribed by Malians tradipraticians with the local populations in the treatment of the pain: Guiera senegalensis. Initially, an ethnobotanic investigation by the Malian tradipraticians enabled us to choose the places of collection of the material and selecting the parts to be taken on the plant (leaves, bark, roots …). After crushing and various extractions with organic solvents, we obtain several crude extracts whose analgesic activity is evaluated . The active fractions are then purified and the active ingredients isolated and identified in order to determine the most effective amount and inducing the minimum of adverse effects. Moreover, the mechanism of action of the most effective molecules will be studied.

Publications
Somboro A, Patel K, Diallo D, Sidibe L, Chalchat JC, Figueredo G, Ducki S, Troin Y, Chalard* P Journal of Medicinal Plant Research 2011, 5, 1639-1651 : “An ethnobotanical and phytochemical study of the African medicinal plant Guiera senegalensis J. F. Gmel “.Chalard P, Patel K, Caldefie-Chezet F, Delort L. Brevet : FR 2011 58256, “Nouveau principe actif extrait de Guiera senegalensis à activité anticancéreuse”.
Grants
Bourse filière du Conseil Régional d’Auvergne / FEDER (2010-2011)
Ambassade de France de Bamako au Mali
Academic and industrial collaborations
Département de Médecine Traditionnelle (DMT) et la Faculté de Sciences et Technique (FAST) de Bamako, Mali.
